WHEN the DNA sequence of the human genome was revealed in 2000, many people expected it to start a revolution. Researchers would be able to discover the genes that caused or influenced diseases. And drug companies would be able to use that knowledge to create better medicines. Until recently, though, it has been a case of “revolution postponed”. The flood of promised discoveries has been more like a trickle.
Much of the reason for the unfulfilled promises was naivity about how straightforward the link between different versions of genes and particular diseases would be. But that naivity has gone, and the fact that complex illnesses often have contributions from large numbers of genes is now recognised. This recognition, plus better computing and sequencing power, mean researchers are indeed beginning to pick the relationships between genes and disease apart.
In January, for example, a group at the Broad Institute in Cambridge, Massachusetts said they had homed in on most of the genes involved in schizophrenia, and thus had a hypothesis for a mechanism that might be causing it. This week it is the turn of breast cancer, as the most comprehensive analysis yet of mutations related to this condition is published in Nature.
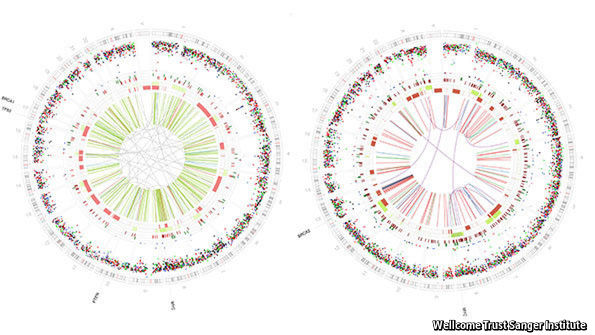
The team that did the analysis, led by Serena Nik-Zainal of the Sanger Institute in Cambridge, England, sequenced the genomes of cells from 560 tumours. These, says Dr Nik-Zainal, proved vastly different from the genomes of healthy cells from the patients involved, for they had generally acquired thousands of mutations. That is not surprising. Early mutations in the development of a tumour often involve genes involved in DNA repair. Once this has happened other mutations accumulate. But all these secondary mutations make it hard to sift out the ones which are clinically relevant. To do so you need to compare lots of samples from different people, and thus see which mutations some of them have in common.
Source: Encore une fois | The Economist